Urgent Recall Notice for Prescription Medication Due to Serious Defect
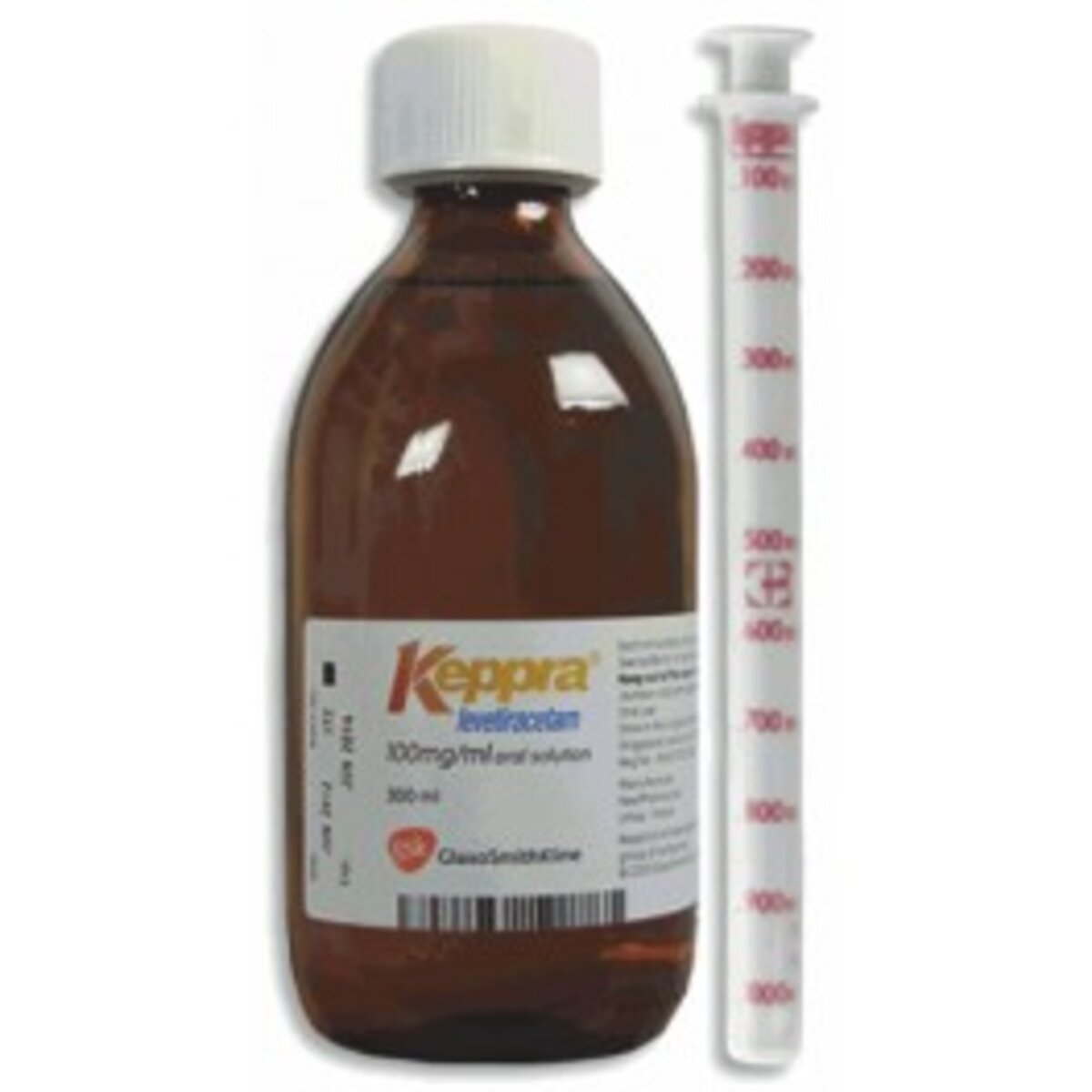
In a recent health alert, the National Public Health and Pharmacology Center (NNGYK) has announced an immediate recall of specific batches of the prescription medication Keppra 100 mg/ml oral solution, due to a significant quality issue.
The affected batch numbers are 23I15 and 23J10.
Authorities have ordered the withdrawal of this product from the market and are calling for its return from healthcare providers and patients alike. The NNGYK's directive includes the replacement of the collected product at pharmacies, ensuring that patients have access to safe medication without delay. The financial responsibilities for the recall, withdrawal, and any suspension of sales fall upon the marketing authorization holder of the product.
The reason behind this decisive action stems from a quality defect identified in the 3 ml syringes used for dosing the Keppra 100 mg/ml oral solution. Specifically, the red ink denoting volume measurements on these syringes is prone to flaking off. This issue was flagged to the NNGYK through the European Rapid Alert System.
Considering the assessment by the Belgian Authority acting as the rapporteur, the visibility issue posed by the missing ink markings could represent a grave danger to patients with epilepsy. The compromised functionality of the dosing device due to this quality defect presents a significant risk, leading to the recall decision for the batches 23I15 and 23J10 of the Keppra 100 mg/ml oral solution (EU/1/00/146).
Patients currently in possession of Keppra oral solution from the specified batches are urged to return them to their pharmacy for a necessary exchange. This measure is critical to ensuring the continued safety and care of epilepsy patients reliant on this medication.
Authorities have ordered the withdrawal of this product from the market and are calling for its return from healthcare providers and patients alike. The NNGYK's directive includes the replacement of the collected product at pharmacies, ensuring that patients have access to safe medication without delay. The financial responsibilities for the recall, withdrawal, and any suspension of sales fall upon the marketing authorization holder of the product.
The reason behind this decisive action stems from a quality defect identified in the 3 ml syringes used for dosing the Keppra 100 mg/ml oral solution. Specifically, the red ink denoting volume measurements on these syringes is prone to flaking off. This issue was flagged to the NNGYK through the European Rapid Alert System.
Considering the assessment by the Belgian Authority acting as the rapporteur, the visibility issue posed by the missing ink markings could represent a grave danger to patients with epilepsy. The compromised functionality of the dosing device due to this quality defect presents a significant risk, leading to the recall decision for the batches 23I15 and 23J10 of the Keppra 100 mg/ml oral solution (EU/1/00/146).
Patients currently in possession of Keppra oral solution from the specified batches are urged to return them to their pharmacy for a necessary exchange. This measure is critical to ensuring the continued safety and care of epilepsy patients reliant on this medication.
Translation:
Translated by AI